Unraveling the dynamics of the processes occuring in a living cell can be achieved by perturbing the system and observing its response. In the preceding years, we introduced efficient photoactivation strategies to introduce perturbations with light as a trigger.[1] During these studies, we used fluorescent probes to report on the system response and became concerned with their limitations in terms of temporal resolution, photostability, and multiplexing opportunities.
Most techniques to visualize a biomolecule of interest in vivo rely on highly fluorescent probes covalently linked to the target biomolecule. The best-known examples are fluorescent proteins, such as the green fluorescent protein that can be genetically fused to a protein of interest. However fluorescent proteins suffer from long maturation of their fluorophore and limited photostability. Moreover the overlap of their absorption and emission spectra permits the simultaneous observation of only three to four fluorescent proteins. To overcome these limitations, it has been proposed to rely on non-covalent labeling with a fluorophore.[2] Hence we recently introduced Yellow Fluorescence-Activating and absorption-Shifting Tag (Y-FAST), a small monomeric protein tag, half as large as the green fluorescent protein, enabling fluorescent labeling of proteins in a reversible and specific manner through the reversible binding and activation of a cell-permeant and nontoxic fluorogenic ligand (a so-called fluorogen) (Figure 1).[3]
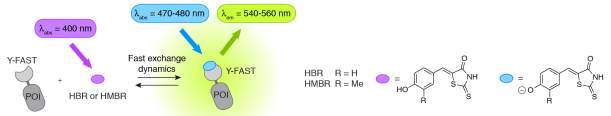
Figure 1. Y-FAST binds the fluorogenic HBR and HMBR and activates their fluorescence (POI: protein of interest). Binding induces two spectroscopic changes: an increase of the fluorescence
quantum yield and an absorption red shift (due to ionization).
A unique fluorogen activation mechanism based on two spectroscopic changes, increase of fluorescence quantum yield and absorption red shift, provides high labeling selectivity. Y-FAST was engineered from the 14-kDa photoactive yellow protein by directed evolution using yeast display and fluorescence-activated cell sorting. Y-FAST is as bright as common fluorescent proteins, exhibits good photostability, and allows the efficient labeling of proteins in various organelles and hosts. Upon fluorogen binding, fluorescence appears instantaneously, allowing monitoring of rapid processes in near real time. Y-FAST distinguishes itself from other tagging systems because the fluorogen binding is highly dynamic and fully reversible, which enables rapid labeling and unlabeling of proteins by addition and withdrawal of the fluorogen (Figure 2), fluorogen exchange before bleaching occurs,[4] and open new prospects for the development of multiplexing imaging protocols based on sequential labeling.
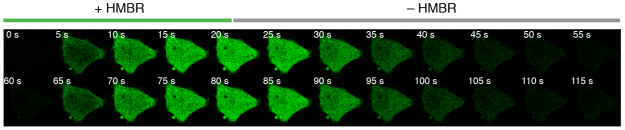
Figure 2. On/off fluorescence switching by iterative labeling/unlabeling. HeLa cells
expressing mCherry-Y-FAST were grown in a microfluidic channel and repeatedly incubated with HMBR-containing culture medium for 20 s and HMBR-free culture medium for 40 s.
Subsequent developments enabled us to introduce a collection of fluorogens for tuning the FAST fluorescence color from green-yellow to orange and red.[5] Beyond allowing the multicolor imaging of FAST-tagged proteins in live cells, these fluorogens permit dynamic color switching because of FAST’s reversible labeling. This unprecedented behavior allows for selective detection of FAST-tagged proteins in cells expressing both green and red fluorescent species through two-color cross-correlation, opening up exciting prospects to overcome spectral crowding and push the frontiers of multiplexed imaging.
References
[1] A. Gautier, C. Gauron, M. Volovitch, D. Bensimon, L. Jullien, S. Vriz, How to control proteins with light in living systems, Nat. Chem. Biol., 2014, 10, 533 – 541.
[2] L. Jullien, A. Gautier, Fluorogen-based reporters for fluorescence imaging : a review, Methods Appl. Fluoresc., 2015, 3, 042007.
[3] M.-A. Plamont, E. Billon-Denis, S. Maurin, C. Gauron, F. M. Pimenta, C. G. Specht, J. Shi, J. Quérard, B. Pan, J. Rossignol, K. Moncoq, N. Morellet, M. Volovitch, E. Lescop, Y. Chen, A. Triller, S. Vriz, T. Le Saux, L. Jullien, A. Gautier, A small fluorescence-activating and absorption shifting tag for tunable protein imaging in vivo, Proc. Nat. Acad. Sci. USA, 2016, 113, 497 – 502.
[4] F. M. Pimenta, G. Chiappetta, T. Le Saux, J. Vinh, L. Jullien, A. Gautier, Chromophore Renewal and Fluorogen-Binding Tags: A Match Made to Last, Sci. Rep., 2017, 7, 12316.
[5] C. Li, M.-A. Plamont, H. L. Sladitschek, V. Rodrigues, I. Aujard, P. Neveu, T. Le Saux, L. Jullien, A. Gautier, Dynamic multicolor protein labeling in living cells, Chem. Sci., 2017, 8, 5598-5605.