Eliminating the contribution of interfering compounds is a key step in chemical analysis. In complex media, one possible approach is to perform a preliminary separation. However purification is often demanding, long, and costly; it may also considerably alter the properties of interacting components of the mixture (e.g. in a living cell). Hence there is a strong interest for developing separation-free non-invasive analytical and imaging protocols.[1] Using photoswitchable probes as labelling and titration contrast agents, we have demonstrated that the association of a modulated light excitation with a kinetic filtering of the overall observable is much more attractive than constant excitation to read-out the contribution from a target probe under adverse conditions (Figure 1).[2-4]
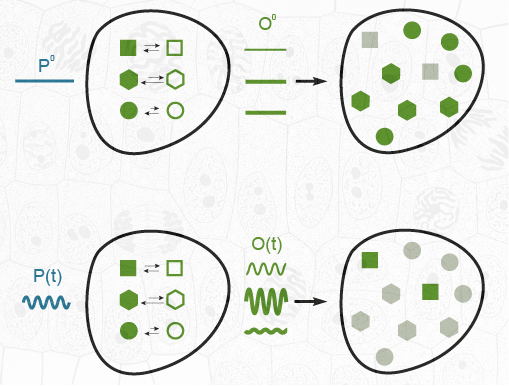
Figure 1. Principle of temporally modulated light excitation and kinetic filtering for selective imaging of photoswitchable probes exchanging between two states of different brightness. Constant illumination reveals all spectrally similar probes of a mixture (top). In contrast, matching the parameters of the modulated illumination (mean intensity, radial frequency of modulation) with the dynamics of a targeted photoswitchable probe permits to selectively retrieve its contribution from the amplitude of the modulated components of the overall signal in the presence of spectrally interfering species, photoswitchable or not (bottom).
An extensive theoretical framework enabled us recently to optimize the out-of-phase concentration first-order response of a photoswitchable probe to modulated illumination by appropriately matching the average illumination and the radial frequency of the light modulation to the probe dynamics. Thus, we can selectively and quantitatively extract from an overall signal the contribution from a target photoswitchable probe within a mixture of species, photoswitchable or not. This simple titration strategy has been more specifically developed in the context of fluorescence micro- and macro-imaging by introducing OPIOM (Out-of-Phase Imaging after Optical Modulation), which offers promising perspectives (Figure 2).
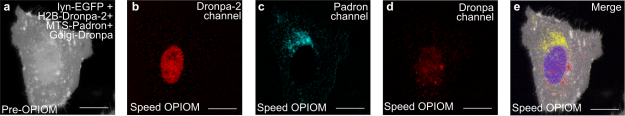
Figure 2. Speed OPIOM can independently image four spectrally similar fluorescent proteins without high contrast enhancement. Pre-OPIOM (a) and Speed OPIOM images tuned to selectively image Dronpa-2 (b), Padron (c), and Dronpa (d). In e, overlay between the pre-OPIOM image from the Dronpa-2 acquisition and Speed OPIOM images collected in b–d. Systems: Fixed U2OS cells expressing H2B-Dronpa-2, Mito-Padron, Dronpa-GTS, and Lyn11-EGFP. Localizations: H2B (nucleus), Lyn11 (cell membrane), Mito (mitochondria), GTS (Golgi). The images were recorded at 37°C. Scale bars: 20 µm.
References
[1] J. Quérard, T. Le Saux, A. Gautier, D. Alcor, V. Croquette, A. Lemarchand, C. Gosse, L. Jullien, Kinetics of reactive modules adds discriminative dimensions for selective cell imaging, ChemPhysChem, 2016, 17, 1396 – 1413.
[2] J. Querard, T.-Z. Markus, M.-A. Plamont, C. Gauron, P. Wang, A. Espagne, M. Volovitch, S. Vriz, V. Croquette, A. Gautier, T. Le Saux, L. Jullien, Photoswitching kinetics and phase sensitive detection add discriminative dimensions for selective fluorescence imaging, Angew. Chem. Int. Ed., 2015, 54, 2633 – 2637.
[3] J. Querard, A. Gautier, T. Le Saux, L. Jullien, Expanding discriminative dimensions for analysis and imaging, Chem. Sci., 2015, 6, 2968 – 2978.
[4] J. Quérard, R. Zhang, Z. Kelemen, M.-A. Plamont, X. Xie, R. Chouket, I. Roemgens, Y. Korepina, S. Albright, E. Ipendey, M. Volovitch, H. L. Sladitschek, P. Neveu, L. Gissot, A. Gautier, J.-D. Faure, V. Croquette, T. Le Saux, L. Jullien, Resonant out-of-phase fluorescence microscopy and remote imaging overcome spectral limitations, Nat. Commun., 2017, 8, 969.